In a Coal-fired Thermal Power Plant, multiple high-pressure boilers are in parallel operation to fulfil the demand for steam and power for the process plant. Normally, the Fuel used is mixed type imported coal and Lignite or Indian coal in different combinations considering the cost economy. In parallel Boiler operations, during variations of coal feed flow due to any trouble in the fuel feeding system or outage of any boiler, an operator will try to maintain plant load by switching steam load on other boilers working healthy. During such instances, there is a chance of an increase in carbon monoxide (CO) in the flue gas which is a potentially fatal gas.
An accurate measurement of the carbon monoxide (CO) concentration in the boiler flue gas can be used to achieve the goals of combustion efficiency, pollutant emissions reduction, and safe operation. By measuring the concentration of CO, power plants can fine-tune the air-to-fuel ratio used on the burners to obtain the highest combustion efficiency. Measuring the CO concentration allows the power plants to reduce the amount of combustion air used while ensuring complete combustion, reducing the production of the pollutant NOx. The concentration of CO in the flue gas is also the most sensitive indicator of unburned combustibles in the process and can indicate the emergence of an unsafe situation.
If a higher concentration of carbon monoxide (CO) in the flue gas leaves the boiler and passes through the Electrostatic Precipitators (ESP), where discharge electrodes are highly charged with DC voltage causing sparks. Carbon monoxide (CO) is a flammable gas and its autoignition temperature is around 605°C. An excessive amount of carbon monoxide (CO) is present in the flue gas, which may lead to creating an uncontrolled explosion in the ESP. which is a very dangerous phenomenon for the boiler, ESP and surroundings.
Analysis:
During an interruption in coal feeding or outage of any of the boilers from running in parallel operation, the panel operator will try to recover boiler firing and maintain bed temperature by increasing the coal feeding to match the plant steam and power demand. In such cases, the excess oxygen level in the boiler will go down. Reduction in excess oxygen percentage in the boiler combustion gases and if continued to operate the boiler at a lower excess oxygen level will result in the formation of a high concentration of carbon monoxide (CO) in the flue gas circuit.
A properly kept, efficiently operating boiler produces extremely little amounts of CO, but a badly maintained boiler can produce lethal levels. Carbon monoxide is always present to some extent in the flue gas of boilers, irrespective of the fuels involved or the stage or type of fire. CO results from incomplete combustion, which can arise due to the following:
- Insufficient heat is available in the combustor, i.e. Low Bed Temperature.
- Quenching of the flame reactions, trying to restart the boiler after the hot box up the stage or trying to switch on coal feeding before achieving the required bed temperature.
- Insufficient oxygen, due to insufficient heat/temperature oxygen level will be getting lowered.
The reaction of oxygen and Carbon in the combustion chamber is as:
C + O2 = CO2
CO2 + C = 2CO
In absence of O2, Carbon will take away oxygen from generated CO2 and increase CO formation faster. Carbon monoxide (CO) correlates with the oxygen content in the flue gasses, low excess oxygen increases CO formation. The higher the air ratio and the better the mixing, the lower the CO emission. Operating with a high furnace temperature and a long residence time decreases CO emissions. Because carbon monoxide (CO) emission behaves in a similar way to many other hydrocarbon emissions, it is often used for regulatory purposes as a signal for the overall efficiency of combustion. Thus, regulating CO is often associated with imposing restrictions on hydrocarbon emissions.
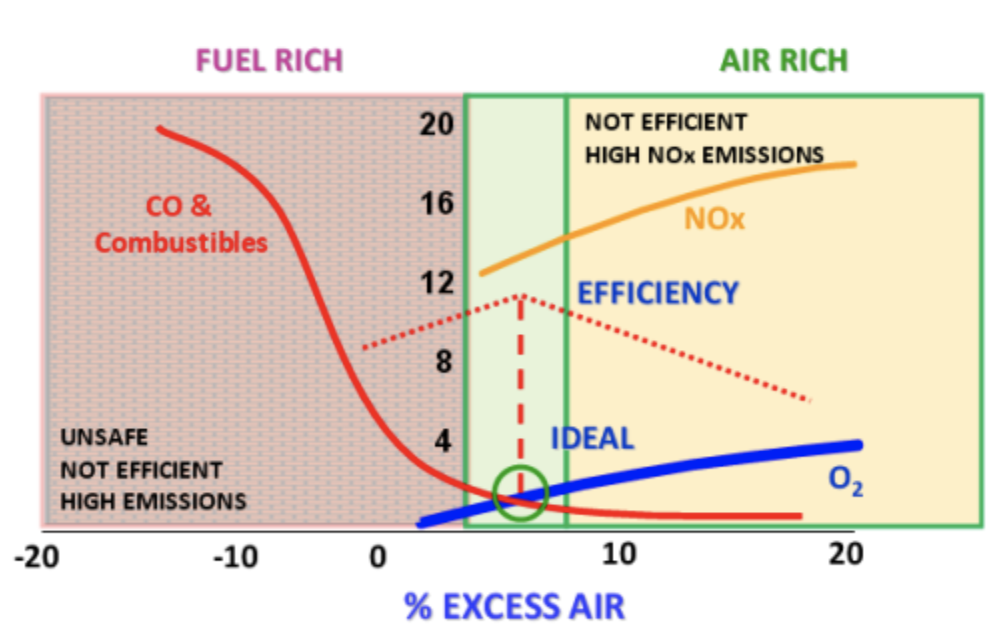
Given complete mixing, a precise or stoichiometric amount of air is required to completely react with a given quantity of fuel to produce complete combustion. In reality, conditions are never ideal so additional or excess air must be supplied to completely burn the fuel. Too little excess air will result in a fuel-rich situation producing a flue gas containing unburned combustibles (carbon monoxide, soot, smoke, coal). This situation results in a loss of efficiency because not all of the potential energy of the coal is captured in the combustion process resulting in fuel waste. Combustion processes that run fuel rich create an increase in pollutant emissions. Also, this is not a safe situation as the unburnt fuel could come into contact with an ignition source further down the process resulting in an uncontrolled explosion.
In combustion applications, such as boilers, furnaces, and heaters, CO measurement can be used with temperature and oxygen content to evaluate efficiency and burner performance. To tune a boiler for maximum efficiency and fuel savings, it is recommended carbon monoxide (CO) detector and interlock system along with Oxygen to trim excess O2 levels in boilers. The amount of excess air in the flue gas is determined by measuring the concentration of oxygen (O2). The ideal excess O2 level (the lowest possible that allows complete combustion) depends on several factors: the fuel type, the burner type, humidity changes in the air, moisture content changes in the fuel, varying boiler loads, fouling of the burner system, and mechanical wear of combustion equipment. Since many of these factors are continuously changing, the ideal amount of excess oxygen continuously changes as well. Measuring carbon monoxide (CO) can help to determine the excess oxygen setpoint in varying load conditions.
Carbon monoxide (CO) is the most sensitive indicator of incomplete combustion. As the amount of excess O2 is reduced, the emergence of CO will occur before other combustibles appear (unburnt fuel). When the concentration of CO reaches the desired setpoint (typically around 100-200 ppm according to boiler design, fuel used, boiler load, etc), the excess O2 concentration is at the desired level and needs to change the excess O2 setpoint. As the concentration of CO increases or decreases, the excess O2 setpoint can be trimmed accordingly. CO trim control of excess O2 concentration assures minimal energy loss, maximum efficiency, and reduced NOx emissions independent of boiler load, fuel type, humidity, moisture content of the fuel and other variables that make excess O2 control difficult. The key to obtaining these benefits is an accurate and reliable measurement of CO in low ppm levels.
ESP OVERVIEW:
ESP has a gas inlet and gas outlet duct. The gas stream entering the ESP passes through perforated screens and is distributed evenly to the interior of the ESP; particulates entrained within the gas stream are consequently also distributed evenly within the ESP.
Discharge electrodes consist of a series of wires that are arranged horizontally across the ESP and installed in several rows. Each discharge electrode is connected to a high voltage supply, which is fed from an electrical system located on top of the ESP housing. Electrical transformers increase the primary supplied high voltage to several thousand volts usually between 20 kV to 70 kV.
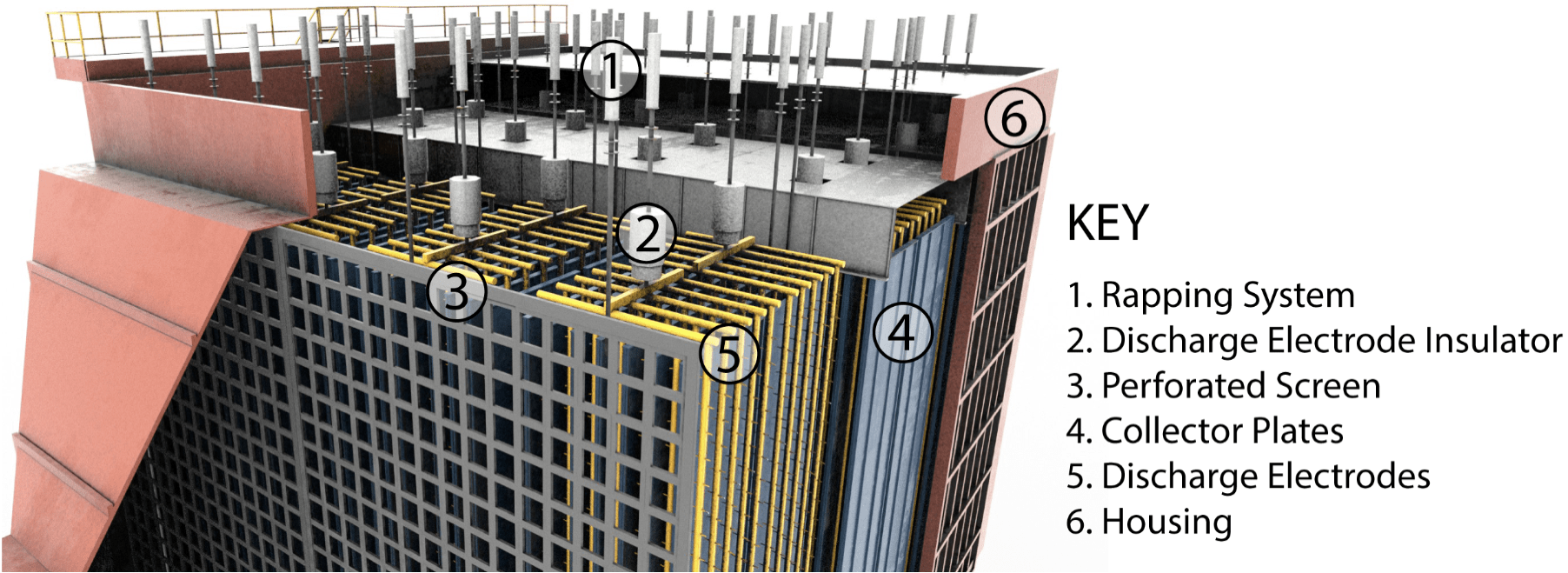
The electrical system incorporates a rectification unit to transform AC voltage to DC voltage. This transformation of AC to DC voltage is necessary to achieve the required electric field that will ionise the particulates as they pass through the ESP. DC voltage is fed to the discharge electrodes, which results in a negative electric field being generated around them. The negative electric field around the discharge electrodes causes a negative charge to be imparted onto the particulates, which causes them to be attracted to the positively charged collector plates. Particulate matter is attracted to the plates by electrostatic force. Once particulate has accumulated on the plates, there is a mechanism for shaking the plates, which causes the particulates to fall due to gravity into the collection hoppers at the base of the ESP and then will be disposed of with a suitable device.
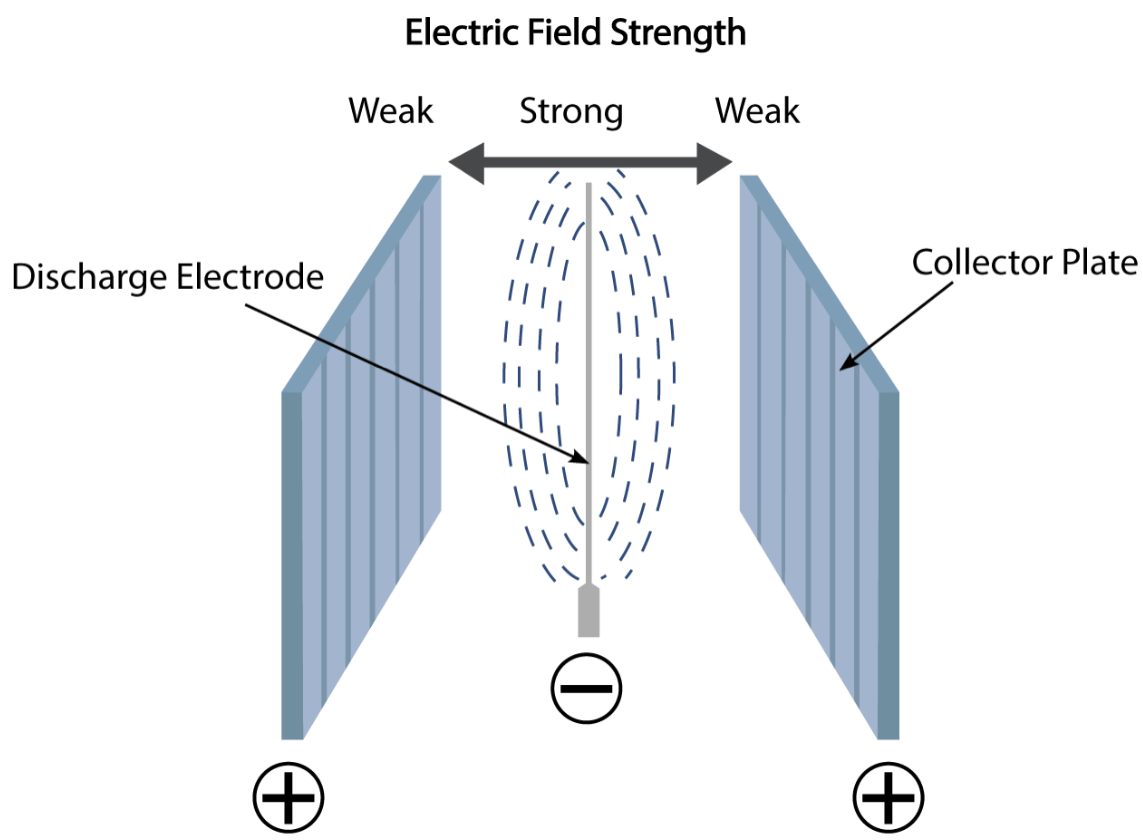
Electrostatic Force
Flue gases with a high concentration of highly flammable carbon monoxide (CO) when reaching the ESP and coming in contact with a high voltage spark will create an uncontrolled explosion in the ESP, and create heavy damage to the boiler, ESP and surroundings.
Conclusions:
- An online Carbon monoxide (CO) detector will help improve the combustion efficiency of the boiler, and trimming excess O2 % level in the boiler will reduce CO generation. Auto combustion based on CO concentration will help improve boiler efficiency.
- Maintain oxygen level in the boiler with sufficient excess air according to the fuel used.
- ESP Operation stop logic can be incorporated in case of lower oxygen level i.e. ESP tripping with < 1-2% of oxygen to safeguard the Boiler & ESP from the explosion due to excessive carbon monoxide (CO).
References:
- ESC BOILER TECHNOLOGIES – http://cleanboiler.org/learn-about/boiler-efficiency-improvement/boiler-combustion/
- https://www.yokogawa.com/us/library/resources/application-notes/co-measurement-in-coal-fired-plants/
- savree.com/en/encyclopedia/electrostatic-precipitator-esp
Anil D. Vyas, Founder, ADV PowerCon, Energy & Power Plant Consultancy Services


Mahendra
September 6, 2022 at 7:42 amExcellent
Abhimanyu Pradhan
September 7, 2022 at 8:16 amNice information sir.